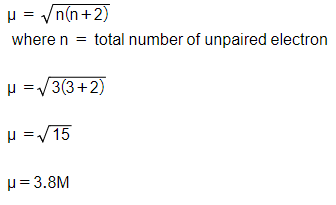
Calculate the spin only magnetic moment of M2+(aq) ion (Z = 27). from Chemistry The d-And-f-Block Elements Class 12 CBSE

The highest value of the calculated spin-only magnetic moment (in BM) among all the transition - YouTube

The spin-only magnetic moment [in units of Bohr magneton, (mu(B) of Ni^(2+) in aqueous solution would be (atomic number of Ni=28)
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84BM. The correct one is - Sarthaks eConnect | Largest Online Education Community
Calculate the spin only magnetic moment of La^3+. - Sarthaks eConnect | Largest Online Education Community

SOLVED: Calculate the magnetic moment of V 3+, Cr3+, Pr3+, Nd3+ according to the following instructions. (a) Consider spin-only magnetic moment in your calculation (b) Consider both spin and orbital moment in
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84 BM. The correct one is - Sarthaks eConnect | Largest Online Education Community







![Spin only magnetic moment of the compound Hg[Co(SCN)4] is : Spin only magnetic moment of the compound Hg[Co(SCN)4] is :](https://d1hhj0t1vdqi7c.cloudfront.net/v1/VzQyTnk4eDZrYms=/sd/)
![PG.CHEMEASY: Calculate the spin only magnetic momentum µ of K3 [ Mn(CN)6] compound . PG.CHEMEASY: Calculate the spin only magnetic momentum µ of K3 [ Mn(CN)6] compound .](https://1.bp.blogspot.com/-Hj8xwf4KI1c/XVFVbdd76zI/AAAAAAAABHM/wsOjo_b2vak1p56suGIQIGFsF7oRBd7QACLcBGAs/s1600/045%2B-%2BCopy%2B%25282%2529.png)







